 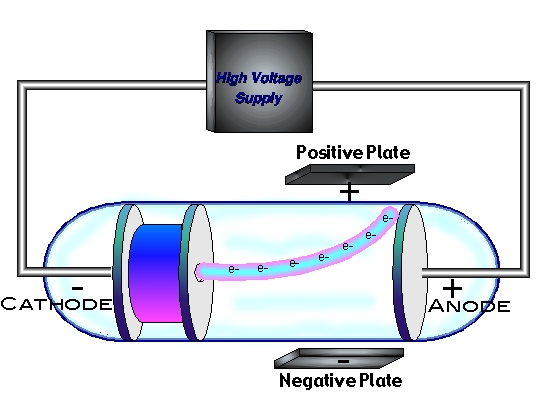
The densely-packed center of the atom is called the nucleus and represents 99.99% of an atom's mass. ➞ reflected particles had a "direct hit" with the center. ➞ deflected particles are those that had come "close to" the positively-charged center. ➞ most alpha-particles pass directly through because the atom is mostly open space. Rutherford's experiments involved bombarding various atoms of gold foil with low-energy alpha particles (α-particles): The - rays scattering experiment is explained with diagrams and. = Rutherford's Gold Foil Experimentġ911 - Ernest Rutherford disproved Thomson's "Plum Pudding Model" of the atom and discovered the nucleus. Thomsons plum pudding model is explained but the cathode ray experiment is not mentioned. ➞ The mass of an electron: m e = 9.1 x 10 -31 kg When the two metal plates are connected to a highvoltage source, the negatively charged plate, called the cathode, emits an invisible ray. ➞ The charge of an electron: 1.6 x 10 -19 C Thomson and essentially discovered the existence of electrons. = The Charge and Mass of the Electronġ909 - Robert Millikan performed experiments that determined the charge of an electron and the mass of an electron. The Cathode Ray Experiment was performed by J.J. NOTE - We are not mentioning the word "protons" here. ➞ tiny, negative, point-charges ( raisins) arranged in a spherical cloud ( pudding) of positive charge. In his Plum Pudding Model, Thomson assumes the electrons are spread out like raisins in a pudding: He estimated the mass of cathode rays by measuring the heat generated when the rays hit a thermal junction and comparing this with the magnetic deflection of the rays. Thomson's Cathode Ray Tube Experiments inspired him to propose his "Plum Pudding Model" of the Atom. Because atoms were known to be neutral, Thomson reasoned that there must be a positive charge somewhere in the atom as well. He did this by directing a stream of cathode rays through narrow slits into the field between two charged plates, and then measuring the streams deflection. Because the "cathode rays" were deflected away from the negative end of an applied electric field, Thomson postulated that the cathode rays are negatively-charged particles called electrons.Ģ. 
When an outside electric field is applied (image on the right), we can see that the electrons (cathode rays) are attracted to the positive end of the applied electric field.ġ. 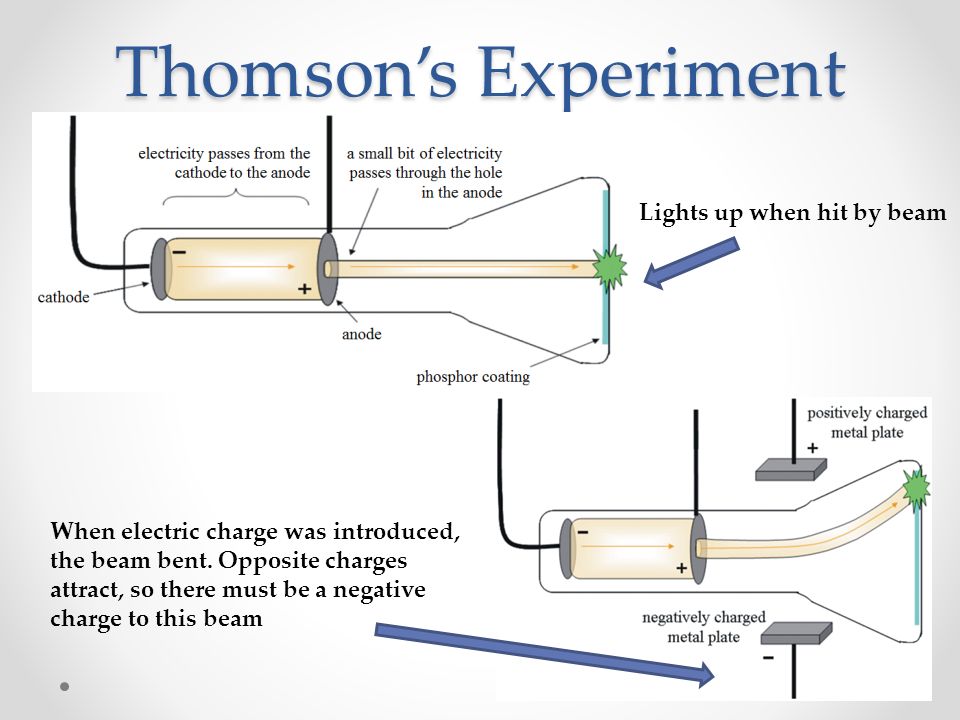
These cathode rays are produced by the applied voltage between the two electrodes. Thomson's Cathode Ray TubeĪs you can see in the image below, cathode rays are streams of electrons running through the cathode ray tube from cathode to anode. Thomson as he conducted experiments with a cathode ray tube. The Electron = discovered in 1903 by J.J. By measuring the deflection and knowing the strength of the electric and magnetic fields, Thomson was able to calculate the charge-to-mass ratio of the electron: $\boldsymbol$.3 - Chemical Quantities and StoichiometryĤ - Types of Chemical Reactions and Solution Stoichiometryħ - Quantum Mechanical View of the Atom, and Periodicityĩ - Covalent Bonding and Molecular Orbitalsġ0 - Liquids, Solids, and Intermolecular Forcesġ5 - Applications of Acid-Base Equilibriaġ6 - Spontaneity, Entropy, and Free Energyġ8 - Transition Metals and Coordination ChemistryĢ0 - An Introduction to Organic Chemistryġ - Structural, Bonding, Molecular PropertiesĢ - The Nature of Organic Compounds: Alkanes and Cycloalkanesģ - Stereochemistry of Alkanes and Cycloalkanes: 3-D Structures of MoleculesĤ - The Study of Organic Reactions: An Overviewġ0 - Substitution (SN2, SN1) and Elimination (E2, E1) Reactionsġ1 - Mass Spectrometry and IR Spectroscopyġ3 - Conjugated Systems and UV Spectroscopyġ5 - Electrophilic Aromatic Substitution (E.A.S.)ġ8 - Aldehydes and Ketones: Nucleophilic Addition ReactionsĢ0 - Carboxylic Acid Derivatives and Nucleophilic Acyl Substitution ReactionsĢ1 - Carbonyl Alpha-Substitution Reactions He observed that the rays were deflected by these fields, indicating that they were charged particles. Thomson's experiment involved applying an electric field and a magnetic field perpendicular to the path of the cathode rays. He discovered that these rays were composed of negatively charged particles, which he called "corpuscles," later known as electrons. 
Thomson conducted a series of experiments using cathode ray tubes to investigate the properties of cathode rays. Thomson's Cathode Ray Tube Experiment and Atom Model: 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
